Transmission Electron Microscopy
Contents |
Overview
The Transmission Electron Microscopes (TEM), similar to Scanning Electron Microscopes (SEM), operates on the same basic optical principles as the light microscope, but uses electrons instead of visible light. The wavelength of visible light limits the resolution of the images able to be produced with a light microscope. Electrons have a much smaller wavelength and therefore enable the TEM to investigate features in the microscale to nanoscale regime with a significantly higher resolution. The possibility for high magnifications has made the TEM a powerful tool in fields such as medical biology and materials research.
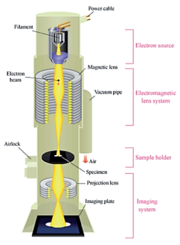
The TEM generates an image by the illumination source (or electron gun) at the top of the microscope emitting electrons that travel through vacuum in the column of the microscope. Instead of glass lenses focusing the light in the light microscope, the TEM uses electromagnetic lenses to focus the electrons into a very thin beam. The electron beam then travels through the thin specimen you want to study. Depending on the density of the material present, some of the electrons are scattered and are dispersed from the beam. At the bottom of the microscope the unscattered electrons hit a fluorescent screen, which produces a "shadow image" of the specimen with its different parts displayed in varied darkness according to their density. The image can be studied directly by the operator or photographed with a camera.
There are a number of drawbacks to the TEM technique. Many materials require extensive sample preparation to produce a sample thin enough to be electron transparent, which makes TEM analysis a relatively time consuming process. The structure of the sample may also be changed during the preparation process. Also the field of view is relatively small, raising the possibility that the region analyzed may not be characteristic of the whole sample. There is potential that the sample may be damaged by the electron beam, particularly in the case of biological materials.
Specimen Preparation
A TEM specimen are required to be at most hundreds of nanometers thick and must be thin enough to transmit sufficient electrons to form an image with minimum energy loss. High quality samples will have a thickness that is comparable to the mean free path of the electrons that travel through the samples, which may be only a few tens of nanometers. Preparation of TEM specimens is specific to the material under analysis and the desired information to obtain from the specimen. As such, many generic techniques have been used for the preparation of the required thin sections. In material science and metallurgy the specimens tend to be naturally resistant to high vacuum inside the TEM that minimizes the possibility of electrons interacting with atoms in the atmosphere prior to coming into contact with the sample. However, these specimens still must be prepared as a thin foil, or etched so some portion of the specimen is thin enough for the beam to penetrate. Constraints on the thickness of the material may be limited by the scattering cross-section of the atoms from which the material is comprised. When two particles interact, the scattering cross section is the area transverse to their relative motion within which they must meet in order to scatter from each other.
For most electronic materials, a common sequence of preparation techniques is ultrasonic disk cutting, dimpling, and ion-milling. Dimpling is a preparation technique that produces a specimen with a thinned central area and an outer rim of sufficient thickness to permit ease of handling. Ion milling is traditionally the final form of specimen preparation. In this process, charged argon ions are accelerated to the specimen surface by the application of high voltage. The ion impingement upon the specimen surface removes material as a result of momentum transfer.
Available at Institute for Imaging and Analytical Technologies
JOEL 2100 200kV TEM
Some features available for the JEOL 2100 200kV scope:
- Lanthanum Hexaboride (LaB6) emitter
- Accelerating voltage - 80,100,120,160,200 kV
- Resolution: Point: 0.23 nm, Lattice: 0.14 nm
- Oxford EDS system for elemental analysis and mapping
- Bottom and side mount Gatan Cameras with Digital Micrograph Software
- Double and single tilt holders (double tilt holders are best suited for rotating to a zone axis in order to obtain Kikuchi patterns in monocrystalline specimens)
- Based on MS Windows™ software
- High-stability goniometer stage and the highest probe current for any given probe size, which allows for improved analytical and diffraction capabilities. The patented JEOL Alpha Selector™ allows a user the selection of a variety of illumination conditions, ranging from full convergent beam to parallel illumination
JEOL 1230 120kV TEM
Some features available for the JEOL 1230 120kV scope:
- Tungsten filament emitter
- Accelerating voltage - 80 kV
- Resolution: Point: .36 nm Lattice: .2 nm
- Bottom mount Gatan Camera with Digital Micrograph Software
- Single tilt holder
- Based on MS Windows™ software
- High-stability goniometer stage
- 5 spot sizes
Experimental Capabilities
Scattered Area Diffraction (SAD) Pattern
Selected area diffraction (SAD) patterns for a specimen are obtained by harnessing the fact that accelerated electrons exhibit wave like properties such as diffraction. Provided that the specimen is of the right thickness, some of the incident electrons will be diffracted similar to a wave passing through a slit. The type of diffraction pattern obtained varies based on the type of material observed. In the case of an amorphous material, the SAD pattern will typically look like a blurry set of concentric circles. On the other hand, crystalline materials show very sharp and discrete SAD patterns owing to the fact that the spacing between atomic planes (which is analogous to the slit width) is fixed for a specific material. Given a small enough SAD aperture diameter and/or a large enough grain size in the crystalline material, the SAD pattern obtained can show discrete spots, with each spot corresponding to electrons that were diffracted between specific planes in a single grain. The symmetry of the diffraction pattern, the distances between the spots, and the relative angles of the spots with respect to the central transmitted beam, can all be used to determine the type of lattice structure, the lattice parameters for the structure, and the angles between each lattice parameter of the unit cell. If the grains are small and/or the aperture is large enough, then the pattern obtained resembles discrete rings. The rings are comprised by a set of discrete spots obtained from each grain . The distances would be the same since the lattice structure is the same for each grain. However, the angular orientation of each grain varies, giving rise to angular rotation of the SAD spots, leading the overall pattern to resemble rings.
Bright Field Imaging (BFI) and Dark Field Imaging (DFI)
Bright field imaging (BFI) is the standard imaging mode used when generating TEM images. A BFI image is generated using the transmitted beam that is captured on a screen or camera. However, some of the incident electrons are diffracted by slight engles, and a dark field imaging (DFI) detector can be used to capture the diffracted electrons and generate a dark field image to potentially highlight features that were not readily visible in the bright field image due to varying contrast levels.
Energy-dispersive X-ray spectroscopy (EDS)
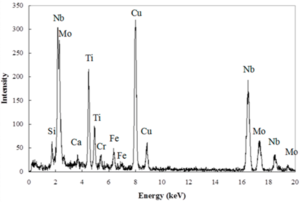
Energy-dispersive X-ray spectroscopy (EDS or EDX) is an analytical technique used for the elemental analysis or chemical characterization of a sample. Its characterization capabilities are due in large part to the fundamental principle that each element has a unique atomic structure allowing a unique set of peaks on its electromagnetic emission spectrum. It relies on the ability to detect X-rays generated when a specimen is bombarded with high-energy electrons in an electron microscope. The X-rays can be detected using an energy-dispersive spectrometer, which can separate X-rays with different energy levels. Qualitative analysis involves the identification of the lines in the spectrum and is fairly straightforward due to the simplicity of X-ray spectra. Quantitative analysis (determination of the concentrations of the elements present) entails measuring line intensities for each element in the sample. All elements from atomic number 4 (Be) to 92 (U) can be detected in principle, though not all instruments are equipped for 'light' elements (Z < 10).
Accuracy
Accuracy of EDS spectrum can be affected by various factors. Many elements will have overlapping peaks. The accuracy of the spectrum can also be affected by the nature of the sample. X-rays can be generated by any atom in the sample that is sufficiently excited by the incoming beam. These X-rays are emitted in any direction, and so they may not all escape the sample. The likelihood of an X-ray escaping the specimen, and thus being available to detect and measure, depends on the energy of the X-ray and the amount and density of material it has to pass through. This can result in reduced accuracy in inhomogeneous and rough samples.