Strain Rate Dependency with Porcine Brain
Contents |
Abstract
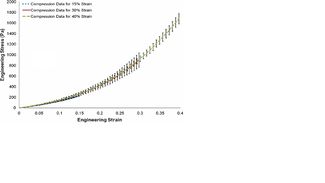
This study examines the internal microstructure evolution of porcine brain during mechanical deformation. Strain rate dependency of porcine brain was investigated under quasi-static compression for strain rates of 0.00625, 0.025, and 0.10s^-1. Confocal microscopy was employed at 15, 30, and 40% strain to quantify microstructural changes, and image analysis was implemented to calculate the area fraction of neurons and glial cells. The nonlinear stress strain behavior exhibited a viscoelastic response from the strain rate sensitivity observed, and image analysis revealed that the mean area fraction of neurons and glial cells increased according to the applied strain level and strain rate. The area fraction for the undamaged state was 7.85 ± 0.07%, but at 40% strain the values were 11.55 ± 0.35%, 13.30 ± 0.28%, and 19.50 ± 0.14% for respective strain rates of 0.00625, 0.025, and 0.10s^-1 . The increased area fractions were a function of the applied strain rate and were attributed to the compaction of neural constituents and the stiffening tissue response. The microstructural variations in the tissue were linked to mechanical properties at progressive levels of compression in order to generate structure–property relationships useful for refining current FE material models.
Methods
Sample Preparation
Intact porcine heads from healthy males were collected from a local abattoir and transported in ice to a necropsy laboratory. Porcine brains were surgically extracted from each skull and temporarily stored in phosphate buffered saline (PBS) to minimize dehydration and tissue degradation. Preparation of test specimens started with a scalpel incision through the corpus callosum in order to separate the left and right hemispheres. A stainless steel die was then used to resect brain material through the sagittal plane of each hemisphere, yielding cylindrical test specimens with the sulci and gyri characterizing the upper surface. The average initial height of the test specimens was 15 mm while the average initial diameter was 30 mm. Brain extraction and dissection required approximately 1 h for completion, and all compression experiments were conducted within 3 h post-mortem.
Testing Apparatus
The Mach-1TM Micromechanical Testing System (BIOMOMENTUM, Quebec), Universal Motion Controller/Driver—Model ESP300, and load cell amplifier were utilized for the compression experiments. The 1.0 kg load cell possessed an error level of ±0.00005 kg and was chosen to adapt to the yielding structural integrity of porcine brain tissue. A circular platen with an estimated diameter of 50mm was also selected to accommodate the smaller cross-sectional area of test specimens. In addition, a stainless steel chamber was fabricated for housing test specimens immersed in 0.01 M PBS in order to minimize tissue dehydration and facilitate the fixing of compressed tissue in 10% neutral buffered formalin (NBF).
Compression Protocol
The compressive strain rates for this study were in the quasi-static range to allow for confocal microscopy and image analysis since the compressed specimens would remain intact for histological processing. In other studies where a split-Hopkinson bar setup was implemented, significantly higher strain rates were achieved to better understand stress wave propagation through brain tissue as observed in high-rate scenarios such as automotive impacts. However, high rate compressed specimens were destroyed beyond recognition, eliminating the possibility for a histological analysis. One of the main objectives in this study was to analyze the fluctuations in the neuron and glial cell arrangement and their correlation to the observed viscoelastic tissue response. The microstructural data could eventually be utilized for assessing the internal changes of neural tissue during the lower threshold of deformation and for evaluating the potential for injury. The strain rate range in this study was chosen based on a similar unconfined compression study on the porcine brain by Miller et al. for strain rates of 0.64, 0.64 x 10^-2, and 0.64 x 10^-5s. The strain rates applied in this study were 0.00625, 0.025, and 0.10s^-1 while the strain levels in which the microstructure was suspended by formalin fixation were 15, 30, or 40%. Each test specimen was compressed once according to the strain rate and strain level designated. The first series of compression experiments were performed to determine if consistent nonlinearity in the stress–strain behavior could be observed when the strain level varied while the strain rate remained fixed. If similar nonlinear stress–strain behavior could be achieved despite variations in the strain levels selected, then confocal images obtained at an intermediate strain (e.g., from brain tissue fixed at 15 or 30% strain) could be assumed to represent the tissue internal microstructure in the compressive loading procedure. In this first series of compression testing at a fixed strain rate of 0.025s^-1, four specimens were compressed to 15% strain, four were compressed to 30% strain, and seven were compressed to 40% strain. The second series of compression experiments were conducted with variations in both the strain rate and the strain level in order to obtain the stress–strain data required to generate structure–property relationships. With the strain level fixed at 40%, 16 specimens were compressed at 0.10s^-1, seven were compressed at 0.025s^-1, and six were compressed at 0.00625s^-1. The original height of each test specimen was determined through a repeatable procedure in which the circular platen was programmed to descend until it initiated contact with the upper surface of the tissue. Contact was confirmed through a load-controlled condition, in which the platen immediately stopped after detecting a load reading of 0.50g. The micromechanical system consisted of a universal displacement actuator platform with a displacement control of 25nm. The built-in actuator sensitivity was utilized for measuring the initial sample height, which was then used for calculating the corresponding loading velocity and the vertical compression for the Mach-1TM programming. A pure slip interface was maintained between the platen surface and the upper tissue surface. While the circular platen remained in a stationary position but still in contact with the test specimen, the chamber was filled with 0.01 M PBS at 25 C to simulate the fluid environment in which the brain is typically surrounded. When full immersion was confirmed, each test specimen was compressed according to the strain rate and strain level specified. Engineering stress and engineering strain were used for data analysis. The cross-sectional area of the specimens was not monitored throughout the entire experiment to avoid tampering with the structural integrity of the tissue. A pre-conditioning step was not incorporated into the testing protocol, because the inherent mechanical integrity of the neural tissue was considered to be incapable of maintaining normal physiological function under cyclic external loading. As a result, a preconditioning was considered to be a contributing factor in the compressive deformation rather than a procedure for tissue preparation. Furthermore, other researchers have elected not to implement any preconditioning steps in their experiments. Toward the end of each experiment, an automated pipette was utilized for siphoning the 0.01 M PBS and replacing it with 10% NBF. Custom-designed polycarbonate cover pieces were immediately positioned over the stainless steel chamber to limit exposure of 10% NBF to the atmosphere while each compressed specimen remained in the fixative for approximately 20–30min. After the preliminary immersion in fixative was complete, each specimen was transferred to a separate storage container filled with a fresh batch of 10% NBF. Stress relaxation was expected to occur because it is an inherent property of viscoelastic tissues such as the brain. Although the fixative was placed on the tissue immediately following compression to 15, 30, and 40%, total fixation did not occur instantly and thus stress relaxation was inevitable to some degree. Stress relaxation was not directly measured in this study, but the authors assumed that the effects were minimal considering that the tissue was undergoing fixation.
Histology
Histology was conducted typically 7–10 days following fixation to provide ample time for sufficient fixative penetration. Rounds of mechanical testing were performed once per week with the intermediate days available for both histology and image analysis of previously tested and fixed tissues.
Image Analysis
ImageJ (National Institute of Health, MD) was utilized for quantifying the microstructural disruption in the neural constituent array within test specimens compressed at different strain rates and strain levels. Selected confocal images were viewed and isolated under CLSM channels corresponding to either the NeuroTrace TM stain or the FluoromyelinTM stain before conversion into 8-bit binary files for image analysis. The ImageJ tools used during the analysis process included outlier removal, threshold adjustment, and particle analysis. The minimum and maximum threshold values were determined by inspection of the analyzed image compared to the original image. This process was repeated if structures other than neurons and glial cells, such as voids, were included in the calculation. Particle analysis was used for outputting multiple parameters, but the main analysis parameter of interest was the area fraction. The area fraction was a quantifiable parameter that defined the overall distribution of the targeted neural constituents, and it was used as a basis of comparison between undamaged tissue and compressed tissue. The differences observed in the area fractions under different compressive testing conditions were intended for use in constitutive modeling, in which the tissue damage could be characterized at the lower threshold of TBI.
Finite Element Modeling
Abaqus/CAE 6.9-1 was utilized for curve fitting the experimental data to existing models found within the commercial finite element code. Separate material databases were generated for each set of compression data and were based on the values for nominal strain and nominal stress recorded. The experimental data for engineering stress vs. engineering strain were used for validating the third order Ogden model (n = 3). The governing equations are shown below, where the terms (li) and (ai) were obtained after using the experimental values for nominal stress and nominal strain: FORMULA HERE where FORMULA HERE Where, in Eqs. (1) and (2), k1, k2, and k3 correspond to the effective principal stretches that are deviatoric in nature. k1 is associated with 1st principal axis (loading direction); k2 and k3 are along the 2nd and 3rd principal axes respectively. J is the determinant of the deformation tensor and Jel is defined as the elastic part of the deformation tensor.