ICME Journal Contribution
Contents |
Abstract
A solute's partial molar volume determines its response to pressure, which can result in changes in molecular conformation or assembly state. Computing speed advances have made accurate partial molar volume evaluation in water routine, allowing for the dissection of the molecular factors underlying this significant thermodynamic variable. A recent simulation analysis of the volumes of nonpolar molecular solutes in water reported that the apparent
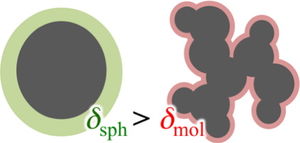
solvent-free border thickness enshrouding these solutes grows with increasing solute size, based on the assumption the solute can be treated as an individual sphere [Biophys. Chem. 161 (2012) 46]. This suggests the solvent dewets these solutes as they grow in size. Via simulations of dewetted repulsive spherical solutes, we show that the border thicknesses of the largest non-polar molecular solutes tend towards that of a repulsive sphere. When attractive interactions are accounted for, however, the spherical solute border thicknesses fall below that of the largest molecular solutes. We demonstrate that if the molecular solutes are treated with atomic detail rather than approximated as an individual sphere, the border thickness variation is minimal. A geometric model is put forward that reproduces the inferred border thickening, indicating the implied dewetting results from a breakdown in the spherical volume approximation.
Author(s): Henry S. Ashbaugh, J. Wesley Barnett, Natalia da Silva Moura, Hayden E. Houser
Introduction
Under the assumption that the hydrated volume of a nonpolar solute can be approximately treated as the packing of water about a sphere, the correlation between partial molar volume and hydrated volume is determined as the solute's partial molar volume less that of an ideal gas particle in solution.
Recent simulations performed by Chalikian and coworkers [1] [2] of a series of solutes composed of atomic Lennard-Jones (LJ) sites with their partial charges turned off to make them nonpolar found that the spherical border thickness grew with increasing solute size. This observation was interpreted as water pulling away from the solute and dewetting the solute/solvent interface as predicted by scaled-particle[3] [4] [5] and Lum-Chandler-Weeks [6] theories. Here we analyze this surprising, and potentially important, result by comparing the measured border thicknesses against simulations of spherical solutes of varying size in water and a simple geometric model to account, in part, for the solute shape.
Methodology
Simulations of individual repulsive, spherical solutes in aqueous solution were performed in the isothermal-isobarric ensemble using the GROMACS 5.0 simulation package [7]. The temperature and pressure were fixed at 25 °C and 1 bar using the Nosé-Hoover thermostat [8]; [9] and Parrinello-Rahman barostat [10], respectively. Water was modeled using the TIP4P/2005 model [11], while solute/water interactions for the repulsive spherical solute was determined by the shifted WCA potential [12], where the position at which the solute/water interaction diverges to infinity has been shifted from the origin to rshift. The WCA potential represents the repulsive portion of the shifted LJ potential following the standard WCA perturbation theory division between attractive and repulsive forces. The LJ well-depth and diameter used here are 3.444 Å and 0.9765 kJ/mol, respectively, corresponding to the cross interactions between water and methane modeled as a united-atom site [13] obtained using Lorentz-Berthelot combining rules [14]. Simulated values of rshift ran from 0 Å to 7 Å in 0.5 Å increments. Simulations were conducted of one solute in a bath of anywhere between 648 and 2307 water molecules depending on the solute size. An additional simulation of 1000 water molecules and no added solute was performed to characterize pure solvent properties. Short range van der Waals interactions were truncated beyond 12 Å with no long-range correction, while electrostatic interactions were evaluated using particle-mesh Ewald summation [15]. Water's internal constraints were held fixed using the SETTLE algorithm [16]. Simulations were conducted for 100 ns following at least 1 ns for equilibration using a time step of 2 fs. Partial molar volumes for the solutes were determined as the difference in the average simulation volume for the system with and without the solute added, with the pure water simulation volume scaled to match the number of waters in the mixture. The ideal gas contribution to the partial molar volume was determined from pure solvent simulation fluctuations to be RTκ0 = 2.02 Å3.
Results
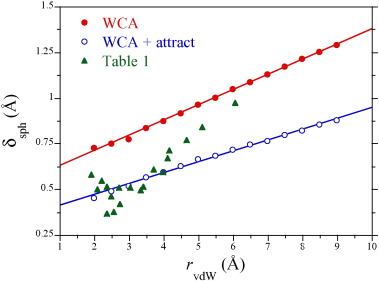
Figure 2 compares the border thicknesses of repulsive, spherical solutes (modeled as shifted Weeks-Chandler-Andersen (WCA) particles) [17] evaluated from simulations in TIP4P/2005 water [18] at 25 °C and 1 atm (see Methods) as a function of their size against those determined by Chalikian and co-workers[19]; [20]for molecularly detailed non-polar solutes. For the repulsive spheres we find that the border thickness is an increasing function of the solute size, consistent with solute dewetting [21]. Dewetting is marked by depression of the water density in the first hydration shell with increasing solute size [22]; [23]; [24]; [25] resulting from an imbalance of forces for solvent molecules partially stripped of their full solvation shell next to a repulsive, low curvature interface [26]; [27].
A shortcoming of the comparison made above is that we neglected attractive solute/water interactions while dispersive interactions were included in the molecular solute simulations composed of LJ sites.
The inference that molecular nonpolar solutes dewet is surprising and stands in difference to successful partial molar volume group contribution correlations, which assume the solute's hydrated volume is the sum of its constituent group volumes without regard to the number of groups making up the solute [28]; [29]; [30]. Indeed, the group volumes largely depend only on local correlations between the group and neighboring water molecules [31]. The growth of the border thickness, however, rests on the assumption that molecular solutes can be approximated as a sphere.
A more realistic correlation for the hydrated solute volume may be obtained by considering the solute's molecular topology explicitly to calculate the border thickness. In this case we evaluate the hydrated volume by adding the border thickness to the van der Waals radii of the constituent atoms that make up the solute (Methods).
A simple model to illustrate how a solute's border thickness can be significantly overestimated is to consider the geometry of spherocylinders. The spherical border thickness for spherocylinders grows by more than a factor of two over than range of solute sizes plotted. Clearly the growth of spherical border thickness for the spherocylinder model is not a result of dewetting since the border thickness is held fixed in this calculation. Rather this indicates a breakdown in the pseudo-spherical description of the solute volume. Indeed, the spherocylinder model predicts spherical border thickness growth in proportion to the effective van der waals radius and does not plateau as might be anticipated for dewetting of a large spherical nonpolar surface [32]; [33].
The accuracy of the spherocylinder model should not be taken as validation that these solutes are cylindrical, which is geometrically unreasonable given the range of molecular structures reported here.
Please see http://dx.doi.org/10.1016/j.bpc.2016.03.002 for the complete article.
References
- ↑ N. Patel, D.N. Dubins, R. Pomes, T.V. Chalikian, "Size dependence of cavity volume: A molecular dynamics study." Biophys. Chem., 161 (2012), pp. 46–49
- ↑ N. Patel, D.N. Dubins, R. Pomes, T.V. Chalikian, "Parsing partial molar volumes of small molecules: A molecular dynamics study." J. Phys. Chem. B, 115 (2011), pp. 4856–4862
- ↑ F.H. Stillinger “Structure in aqueous solutions of nonpolar solutes from the standpoint of scaled-particle theory.” J. Solut. Chem., 2 (1973), pp. 141–158
- ↑ H.S. Ashbaugh, L.R. Pratt “Colloquium: Scaled particle theory and the length scales of hydrophobicity.” Rev. Mod. Phys., 78 (2006), pp. 159–178
- ↑ H.S. Ashbaugh, T.M. Truskett “Putting the squeeze on cavities in liquids: Quantifying pressure effects on solvation using simulations and scaled-particle theory.” J. Chem. Phys., 134 (2011), p. 10
- ↑ K. Lum, D. Chandler, J.D. Weeks “Hydrophobicity at small and large length scales.”J. Phys. Chem. B, 103 (1999), pp. 4570–4577
- ↑ B. Hess, C. Kutzner, D. van der Spoel, E. Lindahl “GROMACS 4: Algorithms for Highly Efficient, Load-Balanced, and Scalable Molecular Simulation”
- ↑ S. Nosé “A Unified Formulation of the constant temperature Molecular-Dynamics Methods.” J. Chem. Phys., 81 (1984), pp. 511–519
- ↑ W.G. Hoover “Canonical dynamics: Equilibrium Phase-Space Distributions.” Phys. Rev. A, 31 (1985), pp. 1695–1697
- ↑ M. Parrinello, A. Rahman “Polymophic Transitions in Single-Crystals - A New Molecular-Dynamics Method.” J. Appl. Phys., 52 (1981), pp. 7182–7190
- ↑ J.L.F. Abascal, C. Vega, “A general purpose model for the condensed phases of water: TIP4P/2005.” J. Chem. Phys., 123 (2005), p. 234505
- ↑ J.D. Weeks, D. Chandler, H.C. Andersen “Role of repulsive forces in determining the equilibrium structure of simple liquids.” J. Chem. Phys., 54 (1971), pp. 5237–5247
- ↑ M.G. Martin, J.I. Siepmann “Transferable potentials for phase equilibria. 1. United-atom description of n-alkanes.” J. Phys. Chem. B, 102 (1998), pp. 2569–2577
- ↑ M.P. Allen, D.J. Tildesley “Computer Simulation of Liquids.” Oxford University Press, Oxford, UK (1987)
- ↑ T. Darden, D. York, L. Pedersen, “Particle mesh Ewald: An N ⋅ log(N) Method for Ewald Sums in large Systems.” J. Chem. Phys., 98 (1993), pp. 10089–10092
- ↑ J.P. Ryckaert, G. Ciccotti, H.J.C. Berendsen, “Numerical-Integration of Cartesian Equations of Motion os a system with constraints - molecular dynamics of n-alkanes.”J. Comput. Phys., 23 (1977), pp. 327–341
- ↑ J.D. Weeks, D. Chandler, H.C. Andersen, “Role of repulsive forces in determining the equilibrium structure of simple liquids.” J. Chem. Phys., 54 (1971), pp. 5237–5247
- ↑ J.L.F. Abascal, C. Vega, “A general purpose model for the condensed phases of water: TIP4P/2005.” J. Chem. Phys., 123 (2005), p. 234505
- ↑ N. Patel, D.N. Dubins, R. Pomes, T.V. Chalikian, “Size dependence of cavity volume: A molecular dynamics study.” Biophys. Chem., 161 (2012), pp. 46–49
- ↑ N. Patel, D.N. Dubins, R. Pomes, T.V. Chalikian, “Parsing partial molar volumes of small molecules: A molecular dynamics study.” J. Phys. Chem. B, 115 (2011), pp. 4856–4862
- ↑ F.M. Floris, “Nonideal effects on the excess volume from small to large cavities in TIP4P water.” J. Phys. Chem. B, 108 (2004), pp. 16244–16249
- ↑ F.H. Stillinger, “Structure in aqueous solutions of nonpolar solutes from the standpoint of scaled-particle theory.” J. Solut. Chem., 2 (1973), pp. 141–158
- ↑ H.S. Ashbaugh, L.R. Pratt “Colloquium: Scaled particle theory and the length scales of hydrophobicity.” Rev. Mod. Phys., 78 (2006), pp. 159–178
- ↑ H.S. Ashbaugh, T.M. Truskett “Putting the squeeze on cavities in liquids: Quantifying pressure effects on solvation using simulations and scaled-particle theory.” J. Chem. Phys., 134 (2011), p. 10
- ↑ K. Lum, D. Chandler, J.D. Weeks “Hydrophobicity at small and large length scales.”J. Phys. Chem. B, 103 (1999), pp. 4570–4577
- ↑ J.D. Weeks, K. Katsov, K. Vollmayr, “Roles of repulsive and attractive forces in determining the structure of nonuniform liquids: Generalized mean field theory.” Phys. Rev. Lett., 81 (1998), pp. 4400–4403
- ↑ G. Hummer, S. Garde, “Cavity expulsion and weak dewetting of hydrophobic solutes in water.” Phys. Rev. Lett., 80 (1998), pp. 4193–4196
- ↑ A.V. Plyasunov, E.L. Shock, “Group contribution values of the infinite dilution thermodynamic functions of hydration for aliphatic noncyclic hydrocarbons, alcohols, and ketones at 298.15 K and 0.1 MPa.” J. Chem. Eng. Data, 46 (2001), pp. 1016–1019
- ↑ G.R. Hedwig, H.J. Hinz, “Group additivity schemes for the calculation of the partial molar heat capacities and volumes of unfolded proteins in aqueous solution.” Biophys. Chem., 100 (2003), pp. 239–260
- ↑ L.N. Surampudi, H.S. Ashbaugh, “Direct evaluation of Polypeptide partial molar volumes in Water using molecular dynamics simulations.” J. Chem. Eng. Data, 59 (2014), pp. 3130–3135
- ↑ A.V. Sangwai, H.S. Ashbaugh, “Aqueous partial molar volumes from simulation, and individual group contributions” Ind. Eng. Chem. Res., 47 (2008), pp. 5169–5174
- ↑ N. Patel, D.N. Dubins, R. Pomes, T.V. Chalikian, "Size dependence of cavity volume: A molecular dynamics study." Biophys. Chem., 161 (2012), pp. 46–49
- ↑ H.S. Ashbaugh, T.M. Truskett “Putting the squeeze on cavities in liquids: Quantifying pressure effects on solvation using simulations and scaled-particle theory.” J. Chem. Phys., 134 (2011), p. 10